an action potential arriving at the presynaptic terminal causes what to occur
Nerve Impulse Transmission within a Neuron: Resting Potential
The resting potential of a neuron is controlled by the divergence in total charge between the inside and exterior of the prison cell.
Learning Objectives
Explicate the formation of the resting potential in neurons
Key Takeaways
Key Points
- When the neuronal membrane is at rest, the resting potential is negative due to the aggregating of more than sodium ions outside the cell than potassium ions inside the prison cell.
- Potassium ions diffuse out of the jail cell at a much faster rate than sodium ions diffuse into the cell considering neurons accept many more potassium leakage channels than sodium leakage channels.
- Sodium-potassium pumps move two potassium ions inside the jail cell every bit three sodium ions are pumped out to maintain the negatively-charged membrane inside the cell; this helps maintain the resting potential.
Key Terms
- ion channel: a poly peptide circuitous or unmarried protein that penetrates a cell membrane and catalyzes the passage of specific ions through that membrane
- membrane potential: the difference in electrical potential across the enclosing membrane of a cell
- resting potential: the nearly latent membrane potential of inactive cells
Nervus Impulse Manual within a Neuron
For the nervous system to role, neurons must be able to send and receive signals. These signals are possible considering each neuron has a charged cellular membrane (a voltage divergence betwixt the inside and the exterior). The charge of this membrane can modify in response to neurotransmitter molecules released from other neurons and environmental stimuli. Whatsoever voltage is a difference in electric potential between two points; for example, the separation of positive and negative electric charges on opposite sides of a resistive barrier. To understand how neurons communicate, ane must first understand the ground of charged membranes and the baseline or 'resting' membrane charge.
Neuronal Charged Membranes
The lipid bilayer membrane that surrounds a neuron is impermeable to charged molecules or ions. To enter or exit the neuron, ions must pass through special proteins called ion channels that span the membrane. Ion channels have different configurations: open up, closed, and inactive. Some ion channels need to exist activated in order to open and allow ions to pass into or out of the cell. These ion channels are sensitive to the environment and tin alter their shape appropriately. Ion channels that alter their structure in response to voltage changes are called voltage-gated ion channels. Voltage-gated ion channels regulate the relative concentrations of different ions inside and outside the prison cell. The difference in total charge between the inside and exterior of the cell is called the membrane potential.

Ion channel configurations: Voltage-gated ion channels are airtight at the resting potential and open up in response to changes in membrane voltage. After activation, they become inactivated for a brief period and volition no longer open in response to a signal.
Resting Membrane Potential
For quiescent cells, the relatively-static membrane potential is known every bit the resting membrane potential. The resting membrane potential is at equilibrium since information technology relies on the constant expenditure of energy for its maintenance. Information technology is dominated past the ionic species in the organisation that has the greatest conductance across the membrane. For most cells, this is potassium. As potassium is also the ion with the about-negative equilibrium potential, normally the resting potential can exist no more negative than the potassium equilibrium potential.
A neuron at residue is negatively charged because the within of a cell is approximately 70 millivolts more negative than the outside (−seventy mV); this number varies past neuron type and past species. This voltage is called the resting membrane potential and is caused by differences in the concentrations of ions inside and outside the prison cell. If the membrane were as permeable to all ions, each type of ion would flow across the membrane and the organization would achieve equilibrium. Because ions cannot but cross the membrane at volition, at that place are different concentrations of several ions inside and outside the cell. The deviation in the number of positively-charged potassium ions (K+) within and exterior the cell dominates the resting membrane potential. When the membrane is at rest, Grand+ ions accumulate within the cell due to a net movement with the concentration gradient. The negative resting membrane potential is created and maintained by increasing the concentration of cations outside the cell (in the extracellular fluid) relative to within the jail cell (in the cytoplasm). The negative charge within the prison cell is created past the cell membrane being more permeable to K+ movement than Na+ movement.
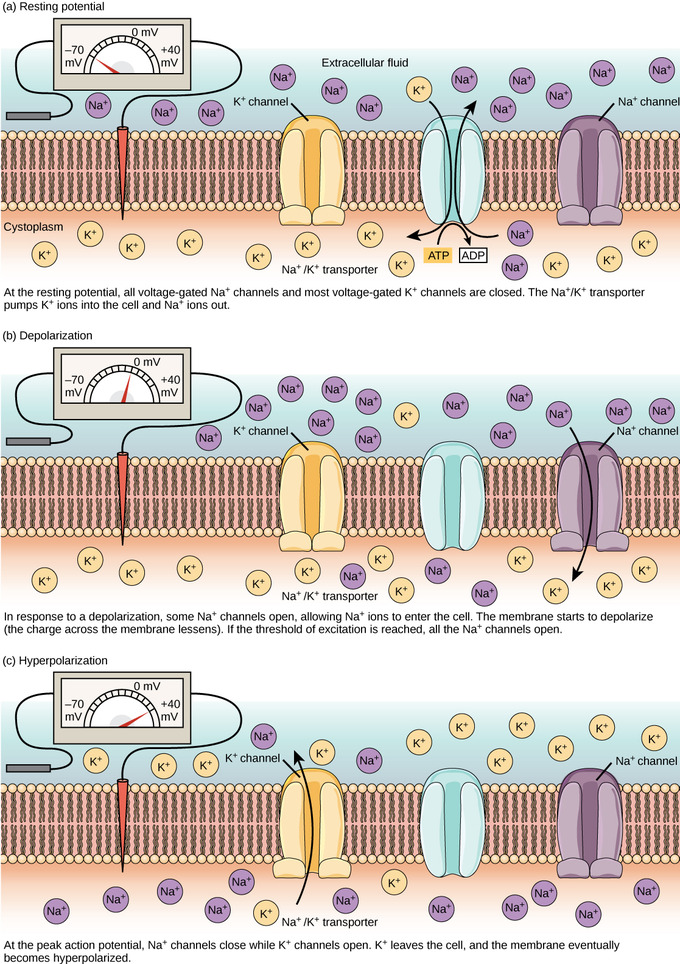
Membrane potential: The (a) resting membrane potential is a result of dissimilar concentrations of Na+ and K+ ions inside and outside the jail cell. A nerve impulse causes Na+ to enter the cell, resulting in (b) depolarization. At the height action potential, K+ channels open and the cell becomes (c) hyperpolarized.
In neurons, potassium ions (K+) are maintained at high concentrations inside the prison cell, while sodium ions (Na+) are maintained at high concentrations outside of the cell. The jail cell possesses potassium and sodium leakage channels that allow the two cations to diffuse downwardly their concentration slope. However, the neurons have far more potassium leakage channels than sodium leakage channels. Therefore, potassium diffuses out of the cell at a much faster charge per unit than sodium leaks in. More cations leaving the cell than inbound it causes the interior of the cell to be negatively charged relative to the outside of the cell. The deportment of the sodium-potassium pump help to maintain the resting potential, once information technology is established. Recall that sodium-potassium pumps bring ii 1000+ ions into the cell while removing three Na+ ions per ATP consumed. As more cations are expelled from the cell than are taken in, the inside of the cell remains negatively charged relative to the extracellular fluid.
Nerve Impulse Transmission within a Neuron: Action Potential
Signals are transmitted from neuron to neuron via an activity potential, when the axon membrane chop-chop depolarizes and repolarizes.
Learning Objectives
Explain the formation of the action potential in neurons
Key Takeaways
Fundamental Points
- Action potentials are formed when a stimulus causes the cell membrane to depolarize past the threshold of excitation, causing all sodium ion channels to open.
- When the potassium ion channels are opened and sodium ion channels are airtight, the cell membrane becomes hyperpolarized as potassium ions leave the cell; the prison cell cannot burn down during this refractory catamenia.
- The activeness potential travels down the axon equally the membrane of the axon depolarizes and repolarizes.
- Myelin insulates the axon to prevent leakage of the current as it travels down the axon.
- Nodes of Ranvier are gaps in the myelin forth the axons; they contain sodium and potassium ion channels, allowing the activity potential to travel quickly downwards the axon past jumping from one node to the next.
Key Terms
- activeness potential: a short term change in the electrical potential that travels along a cell
- depolarization: a decrease in the deviation in voltage between the within and outside of the neuron
- hyperpolarize: to increase the polarity of something, especially the polarity across a biological membrane
- node of Ranvier: a modest constriction in the myelin sheath of axons
- saltatory conduction: the process of regenerating the activity potential at each node of Ranvier
Activity Potential
A neuron can receive input from other neurons via a chemical chosen a neurotransmitter. If this input is strong enough, the neuron will pass the indicate to downstream neurons. Transmission of a bespeak within a neuron (in one direction only, from dendrite to axon terminal) is carried out past the opening and closing of voltage-gated ion channels, which cause a brief reversal of the resting membrane potential to create an activeness potential. As an action potential travels downwardly the axon, the polarity changes across the membrane. One time the signal reaches the axon concluding, information technology stimulates other neurons.

Formation of an action potential: The formation of an action potential tin be divided into five steps. (one) A stimulus from a sensory prison cell or another neuron causes the target jail cell to depolarize toward the threshold potential. (2) If the threshold of excitation is reached, all Na+ channels open and the membrane depolarizes. (3) At the peak action potential, One thousand+ channels open and K+ begins to go out the cell. At the same time, Na+ channels close. (four) The membrane becomes hyperpolarized as K+ ions continue to leave the cell. The hyperpolarized membrane is in a refractory menses and cannot burn down. (5) The Thousand+ channels close and the Na+/Chiliad+ transporter restores the resting potential.
Depolarization and the Action Potential
When neurotransmitter molecules bind to receptors located on a neuron's dendrites, voltage-gated ion channels open. At excitatory synapses, positive ions inundation the interior of the neuron and depolarize the membrane, decreasing the difference in voltage between the inside and outside of the neuron. A stimulus from a sensory cell or some other neuron depolarizes the target neuron to its threshold potential (-55 mV), and Na+ channels in the axon hillock open, starting an action potential. In one case the sodium channels open, the neuron completely depolarizes to a membrane potential of near +40 mV. The activity potential travels downwardly the neuron as Na+ channels open.
Hyperpolarization and Return to Resting Potential
Action potentials are considered an "all-or nil" event. One time the threshold potential is reached, the neuron completely depolarizes. As soon as depolarization is complete, the jail cell "resets" its membrane voltage dorsum to the resting potential. The Na+ channels shut, showtime the neuron'due south refractory menses. At the same time, voltage-gated G+ channels open, assuasive K+ to leave the jail cell. As Thou+ ions exit the cell, the membrane potential in one case again becomes negative. The improvidence of K+ out of the cell hyperpolarizes the jail cell, making the membrane potential more negative than the prison cell's normal resting potential. At this signal, the sodium channels return to their resting country, ready to open again if the membrane potential again exceeds the threshold potential. Eventually, the extra Grand+ ions diffuse out of the cell through the potassium leakage channels, bringing the jail cell from its hyperpolarized country back to its resting membrane potential.
Myelin and Propagation of the Action Potential
For an action potential to communicate information to another neuron, it must travel along the axon and accomplish the axon terminals where information technology tin initiate neurotransmitter release. The speed of conduction of an action potential along an axon is influenced by both the bore of the axon and the axon's resistance to current leak. Myelin acts as an insulator that prevents electric current from leaving the axon, increasing the speed of activeness potential conduction. Diseases like multiple sclerosis crusade degeneration of the myelin, which slows action potential conduction considering axon areas are no longer insulated so the electric current leaks.

Action potential travel forth a neuronal axon: The action potential is conducted down the axon as the axon membrane depolarizes, then repolarizes.
A node of Ranvier is a natural gap in the myelin sheath forth the axon. These unmyelinated spaces are about i micrometer long and contain voltage gated Na+ and K+ channels. The flow of ions through these channels, particularly the Na+ channels, regenerates the activity potential over and over again forth the axon. Activeness potential "jumps" from one node to the next in saltatory conduction. If nodes of Ranvier were not present along an axon, the action potential would propagate very slowly; Na+ and K+ channels would have to continuously regenerate action potentials at every point along the axon. Nodes of Ranvier also salve energy for the neuron since the channels only need to be present at the nodes and not along the entire axon.

Nodes of Ranvier: Nodes of Ranvier are gaps in myelin coverage along axons. Nodes contain voltage-gated K+ and Na+ channels. Activity potentials travel down the axon past jumping from one node to the adjacent.
Synaptic Manual
Synaptic transmission is a chemical effect which is involved in the transmission of the impulse via release, diffusion, receptor bounden of neurotransmitter molecules and unidirectional communication betwixt neurons.
Learning Objectives
Describe the procedure of synaptic transmission
Key Takeaways
Central Points
- In a chemical synapse, the pre and post synaptic membranes are separated by a synaptic cleft, a fluid filled infinite.
- The chemical event is involved in the manual of the impulse via release, diffusion, receptor bounden of neurotransmitter molecules and unidirectional communication between neurons.
- The neurotransmitter termination tin occur in iii ways – reuptake, enzymatic deposition in the cleft and improvidence.
Synaptic Transmission
In a chemical synapse, the pre and post synaptic membranes are separated by a synaptic crevice, a fluid filled space. The chemic outcome is involved in the transmission of the impulse via release, improvidence, receptor binding of neurotransmitter molecules and unidirectional communication betwixt neurons.
Chemical Synapse
Neurotransmission at a chemical synapse begins with the arrival of an action potential at the presynaptic axon terminal. When an action potential reaches the axon terminal, it depolarizes the membrane and opens voltage-gated Na+ channels. Na+ ions enter the cell, further depolarizing the presynaptic membrane. This depolarization causes voltage-gated Ca2+ channels to open. Calcium ions inbound the jail cell initiate a signaling pour. A calcium sensing poly peptide binds calcium and interacts with SNARE proteins. These SNARE proteins are involved in the membrane fusion. The synaptic vesicles fuse with the presynaptic axon last membrane and empty their contents by exocytosis into the synaptic crevice. Calcium is quickly removed from the last.

Synaptic vesicles within a neuron: This pseudocolored image taken with a scanning electron microscope shows an axon terminal that was broken open to reveal synaptic vesicles (blueish and orange) within the neuron.
Fusion of a vesicle with the presynaptic membrane causes neurotransmitters to be released into the synaptic scissure. The neurotransmitter diffuses across the synaptic cleft, bounden to receptor proteins on the postsynaptic membrane.

Advice at a chemical synapse: Communication at chemical synapses requires release of neurotransmitters. When the presynaptic membrane is depolarized, voltage-gated Ca2+ channels open and allow Ca2+ to enter the cell. The calcium entry causes synaptic vesicles to fuse with the membrane and release neurotransmitter molecules into the synaptic cleft. The neurotransmitter diffuses across the synaptic fissure and binds to ligand-gated ion channels in the postsynaptic membrane, resulting in a localized depolarization or hyperpolarization of the postsynaptic neuron.
The binding of a specific neurotransmitter causes item ion channels, in this case ligand-gated channels, on the postsynaptic membrane to open. The bounden of a neurotransmitter to its receptor is reversible. Equally long as it is spring to a post synaptic receptor, a neurotransmitter continues to touch on membrane potential. The effects of the neurotransmitter generally lasts few milliseconds earlier being terminated. The neurotransmitter termination can occur in three ways. First, reuptake by astrocytes or presynaptic terminal where the neurotransmitter is stored or destroyed past enzymes. Second, degradation by enzymes in the synaptic cleft such as acetylcholinesterase. Third, improvidence of the neurotransmitter as information technology moves away from the synapse.
Signal Summation
Signal summation occurs when impulses add together to achieve the threshold of excitation to burn down a neuron.
Learning Objectives
Describe signal summation
Fundamental Takeaways
Primal Points
- Simultaneous impulses may add together together from different places on the neuron to reach the threshold of excitation during spatial summation.
- When individual impulses cannot achieve the threshold of excitation on their ain, they can can add upwards at the same location on the neuron over a short time; this is known every bit temporal summation.
- The action potential of a neuron is fired merely when the net alter of excitatory and inhibitory impulses is non-zero.
Key Terms
- temporal summation: the issue when impulses received at the same identify on the neuron add up
- spatial summation: the effect when simultaneous impulses received at different places on the neuron add up to fire the neuron
- axon hillock: the specialized part of the soma of a neuron that is continued to the axon and where impulses are added together
Point Summation

Signal summation at the axon hillock: A single neuron can receive both excitatory and inhibitory inputs from multiple neurons. All these inputs are added together at the axon hillock. If the EPSPs are strong enough to overcome the IPSPs and attain the threshold of excitation, the neuron volition burn down.
Each neuron connects with numerous other neurons, oftentimes receiving multiple impulses from them. Sometimes, a single excitatory postsynaptic potential (EPSP) is strong plenty to induce an activeness potential in the postsynaptic neuron, but often multiple presynaptic inputs must create EPSPs around the same time for the postsynaptic neuron to exist sufficiently depolarized to fire an action potential. Summation, either spatial or temporal, is the addition of these impulses at the axon hillock. Together, synaptic summation and the threshold for excitation act as a filter so that random "noise" in the system is not transmitted as of import data.
One neuron often has input from many presynaptic neurons, whether excitatory or inhibitory; therefore, inhibitory postsynaptic potentials (IPSPs) tin can cancel out EPSPs and vice versa. The net change in postsynaptic membrane voltage determines whether the postsynaptic cell has reached its threshold of excitation needed to fire an action potential. If the neuron merely receives excitatory impulses, it will likewise generate an action potential. However, if the neuron receives as many inhibitory as excitatory impulses, the inhibition cancels out the excitation and the nerve impulse will cease at that place. Spatial summation means that the effects of impulses received at different places on the neuron add up and so that the neuron may fire when such impulses are received simultaneously, even if each impulse on its own would not be sufficient to cause firing. Temporal summation means that the furnishings of impulses received at the same identify can add upwards if the impulses are received in close temporal succession. Thus, the neuron may burn down when multiple impulses are received, even if each impulse on its ain would non exist sufficient to cause firing.
Synaptic Plasticity
Synapses experience plasticity by strengthening or weakening over time.
Learning Objectives
Distinguish between long-term potentiation and long-term depression
Key Takeaways
Key Points
- Brusque-term synaptic enhancement occurs when the corporeality of available neurotransmitter is increased, while short-term synaptic depression occurs when the amount of vesicles with neurotransmitters is decreased.
- Synapses are strengthened in long-term potentiation (LTP) when AMPA receptors (which bind to negatively-charged glutamate) are increased, allowing more than calcium ions to enter the cell, causing a higher excitatory response.
- Long-term depression (LTD) occurs when the AMPA receptors are decreased, which decreases the corporeality of calcium ions entering the cell, weakening the synaptic response to the release of neurotransmitters.
- The strengthening and weakening of synapses over fourth dimension controls learning and memory in the encephalon.
Fundamental Terms
- long-term potentiation: a long-lasting (hours in vitro, weeks to months in vivo) increment, typically in aamplitude, of the response of a postsynaptic neuron to a particular pattern of stimuli from a presynaptic neuron
- long-term depression: a long-term weakening of a synaptic connexion
- plasticity: the property of neuron that allows information technology to be strengthened or weakened
Synaptic Plasticity
Synaptic plasticity is the strengthening or weakening of synapses over time in response to increases or decreases in their activeness. Plastic change also results from the alteration of the number of receptors located on a synapse. Synaptic plasticity is the basis of learning and memory, enabling a flexible, functioning nervous system. Synaptic plasticity can be either brusque-term (synaptic enhancement or synaptic depression) or long-term. Two processes in particular, long-term potentiation (LTP) and long-term depression (LTD), are important forms of synaptic plasticity that occur in synapses in the hippocampus: a brain region involved in storing memories.

Long-term potentiation and depression: Calcium entry through postsynaptic NMDA receptors tin can initiate two different forms of synaptic plasticity: long-term potentiation (LTP) and long-term depression (LTD). LTP arises when a single synapse is repeatedly stimulated. This stimulation causes a calcium- and CaMKII-dependent cellular cascade, which results in the insertion of more AMPA receptors into the postsynaptic membrane. The next fourth dimension glutamate is released from the presynaptic prison cell, it will bind to both NMDA and the newly-inserted AMPA receptors, thus depolarizing the membrane more than efficiently. LTD occurs when few glutamate molecules bind to NMDA receptors at a synapse (due to a depression firing rate of the presynaptic neuron). The calcium that does flow through NMDA receptors initiates a different calcineurin and protein phosphatase 1-dependent cascade, which results in the endocytosis of AMPA receptors. This makes the postsynaptic neuron less responsive to glutamate released from the presynaptic neuron.
Short-term Synaptic Enhancement and Low
Curt-term synaptic plasticity acts on a timescale of tens of milliseconds to a few minutes. Brusk-term synaptic enhancement results from more synaptic terminals releasing transmitters in response to presynaptic action potentials. Synapses will strengthen for a short time because of either an increase in size of the readily- releasable pool of packaged transmitter or an increase in the amount of packaged transmitter released in response to each action potential. Depletion of these readily-releasable vesicles causes synaptic fatigue. Brusk-term synaptic depression can also arise from postal service-synaptic processes and from feedback activation of presynaptic receptors.
Long-term Potentiation (LTP)
Long-term potentiation (LTP) is a persistent strengthening of a synaptic connection, which can last for minutes or hours. LTP is based on the Hebbian principle: "cells that burn down together wire together. " There are various mechanisms, none fully understood, behind the synaptic strengthening seen with LTP.
One known mechanism involves a type of postsynaptic glutamate receptor: NMDA (North-Methyl-D-aspartate) receptors. These receptors are normally blocked past magnesium ions. However, when the postsynaptic neuron is depolarized by multiple presynaptic inputs in quick succession (either from one neuron or multiple neurons), the magnesium ions are forced out and Ca2+ ions pass into the postsynaptic jail cell. Next, Caii+ ions entering the jail cell initiate a signaling cascade that causes a different type of glutamate receptor, AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid) receptors, to be inserted into the postsynaptic membrane. Activated AMPA receptors let positive ions to enter the prison cell.
Therefore, the next fourth dimension glutamate is released from the presynaptic membrane, information technology will accept a larger excitatory issue (EPSP) on the postsynaptic cell because the bounden of glutamate to these AMPA receptors will allow more than positive ions into the cell. The insertion of boosted AMPA receptors strengthens the synapse so that the postsynaptic neuron is more probable to fire in response to presynaptic neurotransmitter release. Some drugs co-opt the LTP pathway; this synaptic strengthening can lead to addiction.
Long-term Depression (LTD)
Long-term depression (LTD) is substantially the reverse of LTP: it is a long-term weakening of a synaptic connexion. One mechanism known to crusade LTD also involves AMPA receptors. In this situation, calcium that enters through NMDA receptors initiates a different signaling cascade, which results in the removal of AMPA receptors from the postsynaptic membrane. With the decrease in AMPA receptors in the membrane, the postsynaptic neuron is less responsive to the glutamate released from the presynaptic neuron. While it may seem counterintuitive, LTD may be just equally important for learning and memory as LTP. The weakening and pruning of unused synapses trims unimportant connections, leaving only the salient connections strengthened past long-term potentiation.
Source: https://courses.lumenlearning.com/boundless-biology/chapter/how-neurons-communicate/
Post a Comment for "an action potential arriving at the presynaptic terminal causes what to occur"